What is MegaDerm Phalloplasty?
MegaDerm Phalloplasty
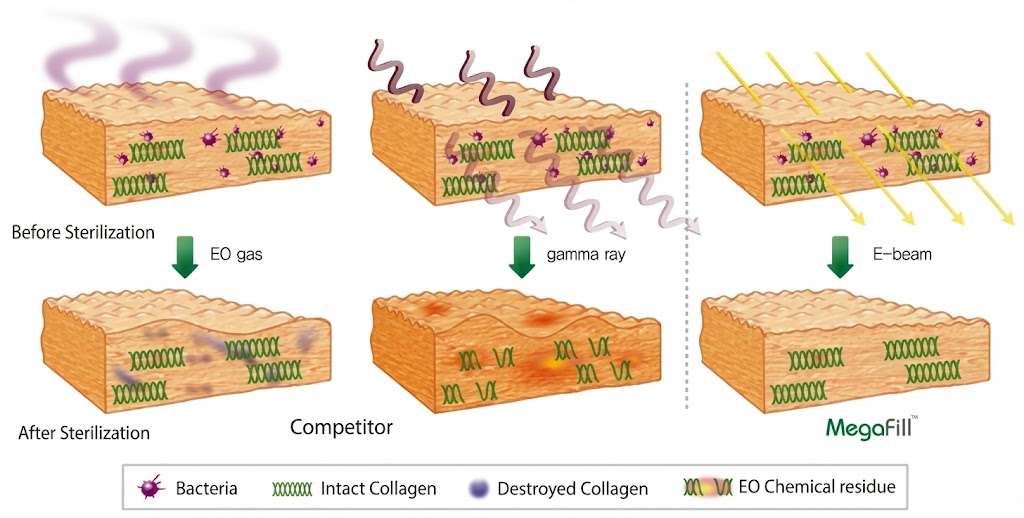
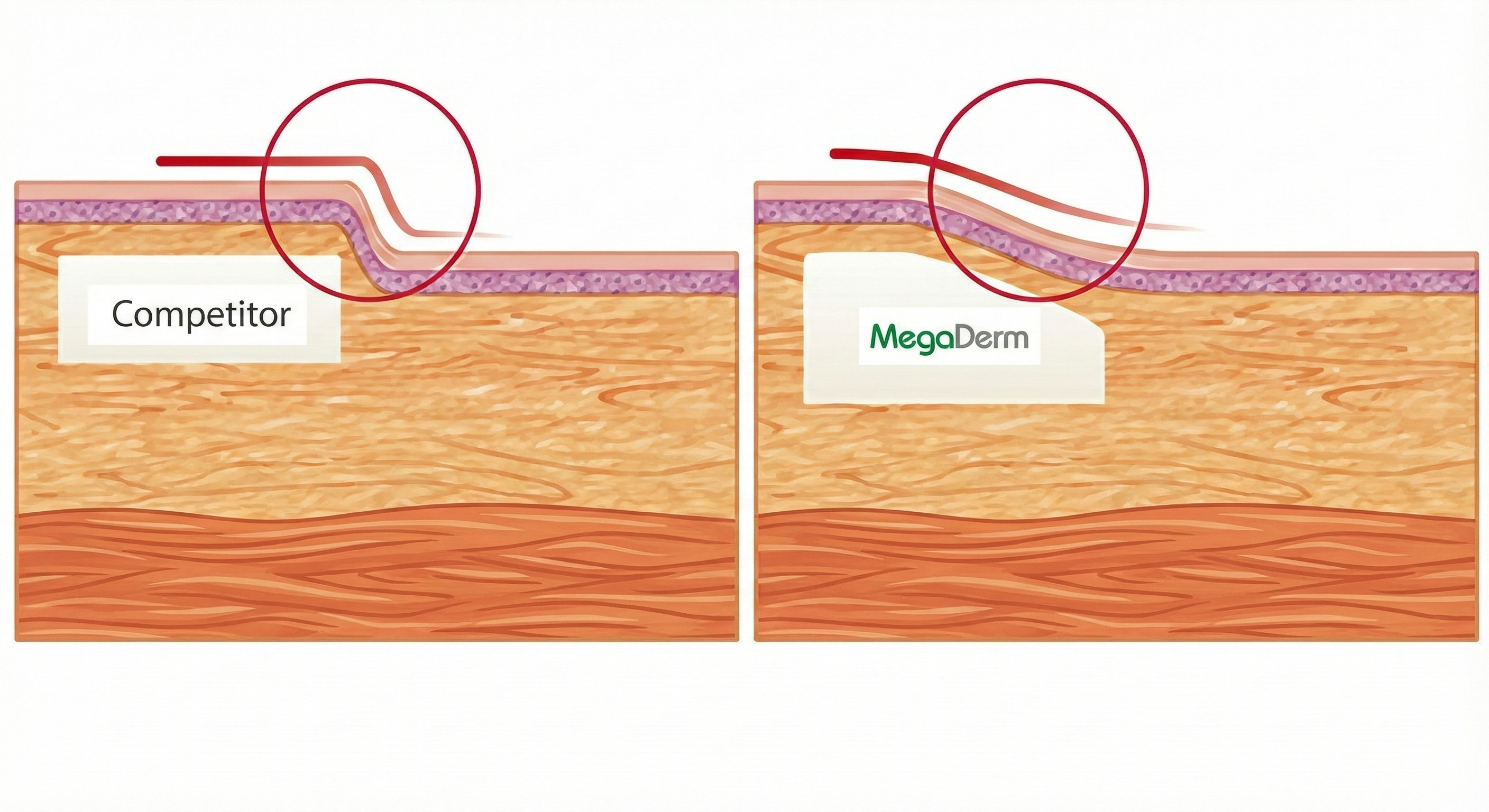
MegaDerm is an acellular dermal matrix allograft sheet offered in Seoul, Korea by Dr Kim Jin Hong. In his opinion, it is as good or better than AlloDerm.
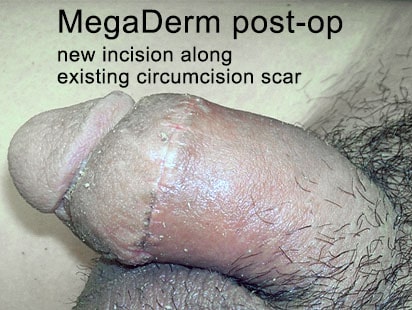
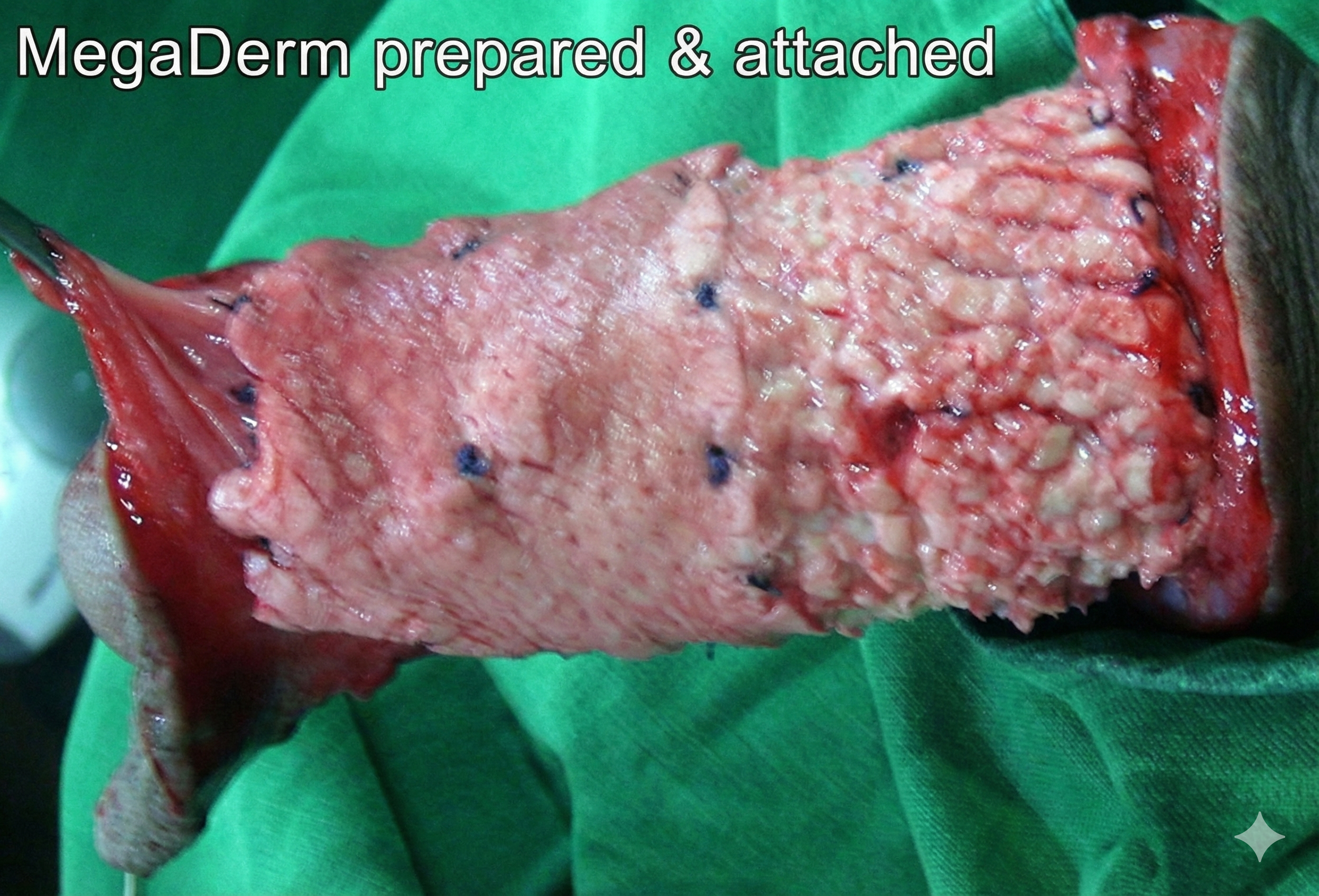
Patients getting this open surgery penile girth enlargement (US$4300 [40 x 70 x 4 mm]) or BOTH glans enhancement (US$2400) and allograft girth enhancement together (US$6700) are advised to stay in Seoul at least 5-6 days to get close and regular aftercare from Dr Kim.
Safety protocols are not negotiable.
Dr Kim says:
"I recommend MegaFill penis enlargement by injection over MegaDerm penis enlargement by surgery."